  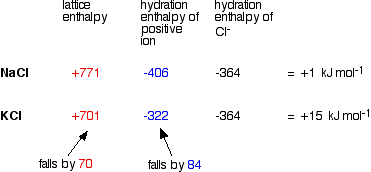
This is 1/5 this is 4/5, and finally, this one here is 1/6. Now we're going to bring everything down. This would be three plus 23 plus two and three plus three. Now, take the periods or rose that they found on the periodic table, and they're gonna go on the bottom, but they're added together. So for all of them, the number will be positive. So here we're gonna multiply their charges in absolute terms. So remember, that's the first part we need for lattice energy. So here all I'm doing is I'm in putting the charges for each one. We're going to have the periods that they're founded. In absolute brackets, we have their charges multiply together on the bottom. Come now, we're gonna say lattice energy for all of them. When you break these all up into their ions, based on the groups that they're found in, we'll be able to figure out the charge here. So remember, highest melting point means highest lattice energy. That should have the highest melting point according to the Ionic bonding model. Remember, the stronger your your lattice energy or high your lattice energy, and the stronger your Ionic bond will be So if you compare the two, we see that magnesium bromide has a larger lattice energy, and therefore it has a stronger ionic bond. Potassium is in the fourth row of the periodic table, so period for chlorine is in the third row. So remember, we multiply their charges in absolute terms. Potassium chloride made up of potassium ion and chloride ion look and see where they are on the periodic table. 
So that is to substance for its lattice energy. So magnesium, if you look on the periodic table is in row three. So plus two times negative one divided by their period numbers added together. Remember, it's their charges in absolute terms. So that breaks up into magnesium two plus NPR minus one for their lattice energy. So for magnesium bromide, we break it up into its ions. Gurney, "Electronic processes in Ionic crystals", 2nd.So here it's saying which compound possesses the strongest Ionic bond? We have magnesium bromide versus potassium chloride. Reguzzoni, Optical properties & electronic structure of alkali halides by thermoreflectivity, Phys. Dutton, Ultraviolet absorption of alkali halides, Phys. Fowler, "Physics of Color Centers", New York: Academic Press (1968) Sugawara Photoelectric emission of Potassium halides in the extreme ultraviolet J. Hamill Anomalous high-field electron injection & photoconduction in thin-film alkali halides, J. Journal de Physique et le Radium, 28 (1981).See also Supplement of information on KCl.The high temperature elastic moduli of alkali halides.Valence band: 0.432 or 0.467 (Hodby) 0.922 for a polaronĮlectron affinity: ( in eV., from bottom of conduction band under vacuum) Pressure & temperature dependence of the static dielectric constants of KCl, LiF & MgO, R.A. Optic dielectric constant: 2.13 or 2.20 (Hodby) Static dielectric constant: 4.68 or 4.49 (Hodby) Mixed approach of linear-combination-of-atomic-orbitals & orthogonalized-plane-wave methods to the band-structure calculation of alkali-halide crystals, S.M. II:Critical survey of theorical calculation, R.T. (Ref.2)Įlectronic band structure of the alkali halides. Longitude optic phonon L0 (k=0): 212 cm -1 (Hodby) which means: 3.99*10 13 rad/sec.Įxciton energy: 8.6 eV. Transverse optic phonon T0 (k=0): 142 or 151 cm -1 Rolandson, Lattice dynamics of NaCl, KCl, RbCl & RbF, Phys.Rev. Stiffness constants: in 10 11 dynes/cm 2, at room temperatureĬompressibility (in 10 11 dynes/cm 2): 0.574 ?
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
